|
ENS(enteric nervous system)
Ever heard your gut called a "second brain"? Well, it's not just a saying. There's a whole neural network down there, the enteric nervous system (ENS), that's basically running its own show. This isn't just about digestion (though it's a pro at that), the ENS is linked to our mood, immune system, and even has some scientists wondering if it affects how we think. So, buckle up, buttercup, because we're about to dive deep into this mind-blowing world of gut intelligence and how it's
shaking up the way we understand our bodies.
This remarkable network of neurons is embedded within the lining of the gastrointestinal tract and plays a critical role in managing digestive processes. Far from being a simple relay system, the ENS operates with a level of autonomy that rivals that of the spinal cord, and it's rich in neurotransmitters like serotonin. As we delve into the functions and impacts of the ENS, we'll uncover its significant influence not only on digestion but also on emotional and mental health. Join us as we unravel
the complexities of this lesser-known but vital part of our nervous system, providing insights into its importance in both physical and psychological well-being.
The enteric nervous system (ENS) isn't located in a single spot like the brain; it's actually woven throughout the entire lining of your gut. It starts in your esophagus and runs all the way down to your rectum. It's made up of two main parts:
- Myenteric Plexus (Auerbach's Plexus): This part is tucked between the muscle layers of your intestines and controls how things move through your digestive system.
- Submucosal Plexus (Meissner's Plexus): This one's found in the submucosa, a deeper layer of your gut wall, and it manages secretions, blood flow, and even talks to your immune system.
NOTE : The term "plexus" comes from the Latin word "plexus," which literally means "braid" or "network." It is related to the verb "plectere," meaning "to weave" or "to twist." This meaning perfectly reflects the structure of a plexus, which is a complex, interwoven network of nerves or vessels. The term is used in anatomy to describe various interconnected structures in the body, such as nerve plexuses (like
the ones in
the enteric nervous system) and blood vessel plexuses.
Think of the ENS like a network of tiny wires sending messages up and down your gut, controlling everything from how fast food moves through to how much stomach acid you produce.
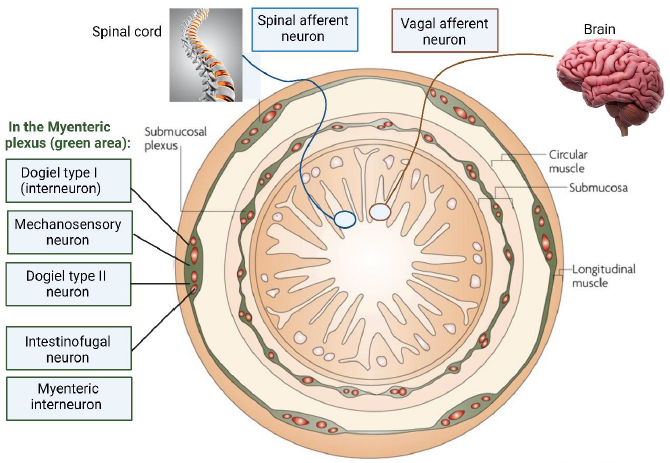
Image Source : Our Mental Health Is Determined by an Intrinsic Interplay between the Central Nervous System, Enteric Nerves, and Gut Microbiota
The illustration shown above depicts the organization of the enteric nervous system (ENS) and its connection to other parts of the nervous system. It depicts various types of neurons and nerve endings within the ENS, particularly focusing on the myenteric and submucosal plexuses, which are two key components of the ENS.
Overall, the image effectively represents the complex network of neurons in the ENS, their location within the gastrointestinal tract, and their functional interactions with both intrinsic and extrinsic nervous systems. This network plays a crucial role in the autonomous regulation of gastrointestinal functions.
- Myenteric Plexus (Green Area): This is located between the circular and longitudinal muscle layers of the gastrointestinal tract. It is highlighted in green and contains two types of neurons:
- Dogiel Type I Neurons: These are mechanosensory and are rapidly adapting and excitatory. They are sensitive to rapid changes but not to tension.
- Dogiel Type II Neurons: These neurons are also mechanosensitive and respond to both rapid synaptic inputs and tension. They are depicted as reacting to both rapid and slow changes in muscle tension.
- Submucosal Plexus: This is located in the submucosa layer of the gastrointestinal tract. It contains interneurons and myenteric motor neurons. These neurons are crucial for regulating the digestive functions such as secretion and absorption within the gut.
- Intestinofugal Neurons: These neurons are termed as "second-order" neurons and react to direct mechanical stimuli. They send signals to the lower gut (distal colon) and are activated by changes in muscle tension.
The enteric nervous system (ENS) may be independent, but it's definitely not a loner. It has a pretty complex relationship with the rest of your nervous system. It interacts with the central nervous system (CNS) and the autonomic nervous system (ANS) through complex pathways that facilitate communication between the gut and the brain. This bidirectional communication system is often referred to as the "gut-brain axis." Here's how the ENS interacts with other parts of the nervous system:
- Autonomic Nervous System (ANS):
- Sympathetic Nervous System: The sympathetic fibers innervate the gastrointestinal tract and primarily exert inhibitory effects on gastrointestinal motility and secretion. These fibers release neurotransmitters like norepinephrine which slow down gut activity during stress or fight-or-flight responses.
- Parasympathetic Nervous System: The parasympathetic fibers, particularly those from the vagus nerve, have a more direct role in stimulating digestive activities. These fibers enhance gut motility and increase secretion by releasing acetylcholine and other neurotransmitters.
- Central Nervous System (CNS):
- Sensory Integration: The CNS receives sensory information from the gut via afferent fibers of the vagus nerve and the spinal afferent neurons. This includes information about the chemical and mechanical conditions of the gut.
- Response and Regulation: The CNS processes this information and sends back signals that can alter digestive functions. These signals are transmitted via efferent pathways in the ANS. For example, the brain can modulate pain perception in the gut and adjust digestive processes based on factors like emotional state or external stress.
- Vagal and Spinal Pathways:
- Vagal Pathways: The vagus nerve is the primary conduit for parasympathetic communication between the brain and the gut. It sends efferent signals to stimulate digestion and receives afferent signals to keep the brain informed about the gut's status.
- Spinal Pathways: Spinal afferent neurons convey sensory and pain signals from the gut to the spinal cord and brain. These pathways help the CNS to monitor and respond to distress or discomfort in the gastrointestinal tract.
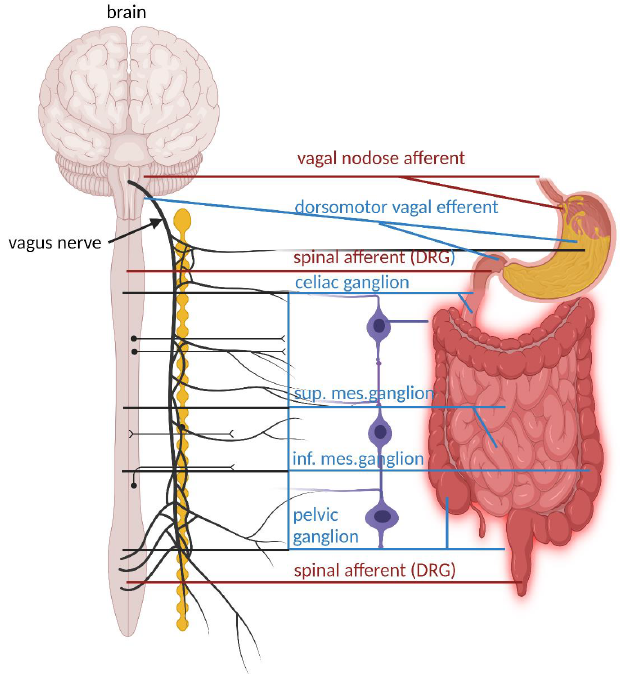
Image Source : Our Mental Health Is Determined by an Intrinsic Interplay between the Central Nervous System, Enteric Nerves, and Gut Microbiota
The illustration show above depicts the major extrinsic neural pathways between the enteric nervous system (ENS), the spinal cord, and the brain. It highlights how the ENS interacts with the central nervous system (CNS) and the autonomic nervous system (ANS) through both afferent (sensory) and efferent (motor) pathways. The diagram captures the complexity of the nerve connections and the significant role of neural communications between the gut and the brain. This gut-brain interaction is critical
for digestive health and also influences emotional and mental well-being, showcasing the profound interconnectedness of our nervous systems.
- Vagus Nerve
- Vagal Nodose Afferent (Red Line): These fibers carry sensory information from the gut to the brain. They detect chemical and mechanical changes in the gut and send signals back to the brain, which are crucial for reflex actions and influencing digestive processes.
- Dorsomotor Vagal Efferent (Blue Line): These are parasympathetic fibers that originate in the brainstem and extend to the gut, where they stimulate digestion by increasing intestinal motility and enzyme secretion.
- Spinal Afferent Neurons (DRG)
- Spinal Afferent Neurons (Red Line): These sensory neurons from the dorsal root ganglia (DRG) provide the CNS with information about the gastrointestinal environment, such as pain and distension. They play a critical role in pain perception and visceral sensations.
- Sympathetic Ganglia
- Celiac Ganglion, Superior Mesenteric Ganglion, and Inferior Mesenteric Ganglion (Blue Circles): These ganglia are part of the sympathetic nervous system and help regulate the gastrointestinal tract by modulating blood flow and gut motility, typically having an inhibitory effect on gastrointestinal secretion and motility.
- Pelvic Ganglion: This ganglion also plays a role in regulating lower parts of the gastrointestinal tract such as the colon and rectum.
- Interaction and Communication:
- Afferent Pathways (Red Lines): Carry sensory information from the gut to the CNS. This information helps the brain monitor and respond to the state of the gastrointestinal tract.
- Efferent Pathways (Blue Lines): Transmit motor commands from the CNS to the gut, controlling various functions such as motility, fluid exchange, and enzyme secretion.
- Parasympathetic and Sympathetic Inputs: The balance between these inputs (parasympathetic generally being excitatory and sympathetic being inhibitory) is crucial for the proper functioning of the gastrointestinal system.
Which one is directly involved in moving gut muscle ? ENS or CNS ?
The Enteric Nervous System (ENS) is the primary system directly involved in controlling the movements of gut muscles. It operates independently within the walls of the gastrointestinal tract and is responsible for regulating peristalsis—the rhythmic contractions that move food through the digestive system. The ENS has its own network of neurons and neurotransmitters, allowing it to autonomously manage digestive processes.
While the ENS handles the direct control of GI muscle movements, it is also modulated by the Central Nervous System (CNS), specifically through interactions with the sympathetic and parasympathetic divisions of the autonomic nervous system. The parasympathetic nervous system, particularly via the vagus nerve, primarily enhances the activity of the ENS by promoting digestion and increasing peristalsis. In contrast, the sympathetic nervous system serves to inhibit the movements of the gut muscles.
It reduces peristalsis and overall digestive activity, especially in stress-related "fight or flight" situations, by signaling through spinal pathways that interact with the ENS.
Thus, while the ENS directly controls the movements of GI muscles, both the parasympathetic and sympathetic branches of the CNS modulate these activities, enhancing or inhibiting them as needed based on the body’s overall physiological state.
What is the difference in terms of functionality between Myenteric Plexus and Submucosal Plexus ?
The myenteric plexus and the submucosal plexus are both essential components of the enteric nervous system (ENS) and play distinct roles in regulating gastrointestinal (GI) functions:
Myenteric Plexus (Auerbach's Plexus):
- Location: Sandwiched between the longitudinal and circular muscle layers of the GI tract.
- Primary Function: Primarily controls GI motility. It regulates the tone, velocity, and intensity of contractions in both muscle layers, coordinating the movement of food along the digestive tract. It is responsible for:
- Peristalsis: The rhythmic waves of contraction that propel food forward.
- Segmentation: The localized contractions that mix food with digestive juices.
- Other Functions: May also play a role in regulating mucosal functions like absorption and secretion to a lesser extent.
Submucosal Plexus (Meissner's Plexus):
- Location: Found within the submucosa, closer to the inner lining of the GI tract.
- Primary Function: Primarily controls local conditions within the gut wall. It regulates secretion of digestive enzymes, mucus, and other substances from the glands in the mucosa. It also influences local blood flow and absorption of nutrients.
- Other Functions: May also have a minor role in controlling smooth muscle contractions in the muscularis mucosae (a thin layer of smooth muscle within the mucosa).
Additional Insights:
- Neurotransmitters and Chemicals: Both plexuses utilize a variety of neurotransmitters such as acetylcholine, serotonin, and nitric oxide, which help in modulating their respective functions effectively.
- Sensory Integration: They also contain sensory neurons that can detect chemical and mechanical conditions within the GI tract, allowing them to adapt their functions in response to different states of digestion and nutrient composition.
Key Differences:
|
Feature
|
Myenteric Plexus
|
Submucosal Plexus
|
|
Location
|
Between muscle layers
|
Within submucosa
|
|
Primary Function
|
Controls GI motility (contraction/movement)
|
Controls local secretion, blood flow, absorption
|
|
Other Functions
|
Minor role in secretion/absorption
|
Minor role in muscle contraction (muscularis mucosae)
|
In summary:
- The myenteric plexus is the "muscle mover," primarily responsible for the coordinated contractions that propel food through the GI tract.
- The submucosal plexus is the "local regulator," controlling secretions, blood flow, and absorption within the gut wall.
Both plexuses work together in a coordinated manner to ensure the smooth and efficient functioning of the digestive system, reflecting a sophisticated level of autonomy and specialized control over the digestive processes.
The enteric nervous system (ENS) interacts extensively with various bodily systems beyond the central and autonomic nervous systems. These interactions highlight the ENS's role as a key player in overall physiological homeostasis and health.
By coordinating these interactions, the ENS not only manages digestion but also plays a significant role in broader health and disease processes. Its ability to integrate neural, immune, and endocrine signals makes it a critical component of body-wide homeostasis and responsiveness to environmental changes.
- Immune System : The ENS directly interacts with the gut-associated lymphoid tissue (GALT), which is part of the mucosal immune system. Neurons in the ENS can influence immune responses by releasing neurotransmitters and neuropeptides that modulate immune cell function within the gut. This is crucial for maintaining gut barrier integrity and managing inflammatory responses, which is important for conditions like inflammatory bowel disease (IBD).
- Endocrine System : The gut is a rich source of hormones, and the ENS plays a crucial role in the regulation of hormone secretion from enteroendocrine cells. These hormones include gastrin, cholecystokinin, and secretin, which regulate aspects of digestion such as enzyme secretion, bile release, and appetite control. The ENS can modulate the release of these hormones in response to food intake and gut distension, integrating neural and hormonal signals to coordinate digestive
processes.
- Circulatory System : The ENS influences blood flow within the gut through its interactions with smooth muscle cells in blood vessels. By regulating vasodilation and vasoconstriction, the ENS ensures adequate blood supply during digestion and nutrient absorption. This is vital for transporting absorbed nutrients to other parts of the body.
- Musculoskeletal System : While the ENS does not directly interact with skeletal muscles, it controls the smooth muscle layers of the gastrointestinal tract. Through coordination of the muscularis externa (which includes layers of longitudinal and circular muscle), the ENS regulates peristalsis— the rhythmic contractions that move food along the digestive tract.
- Respiratory System : Indirectly, the ENS can influence the respiratory system during conditions of distress or discomfort in the gastrointestinal tract, such as acid reflux, which can lead to coughing or changes in breathing patterns due to the close proximity of the digestive and respiratory systems.
- Urinary System : The ENS can affect nearby organs like the bladder through shared neural pathways and local reflexes. For instance, severe constipation can influence bladder function due to mechanical pressure and shared nerve signaling pathways.
- Microbiota : The ENS communicates with the gut microbiota, the vast community of microorganisms living in the intestines. Neurotransmitters and other chemicals released by ENS neurons can influence the behavior and composition of these microbial communities, which in turn can affect nutrient processing, immune modulation, and even neurotransmitter production.
The enteric nervous system (ENS) uses a wide variety of neurotransmitters to regulate gastrointestinal function. These neurotransmitters enable the ENS to communicate within its own network, as well as with the central nervous system (CNS) and other peripheral targets. These neurotransmitters reflect the complexity of the ENS and its capability to handle various aspects of gastrointestinal function, from motility and secretion to blood flow and immune response. Each neurotransmitter has a specific
role and operates within a delicate balance to maintain optimal digestive health.
Most of the neurotransmitters used by the enteric nervous system (ENS) are produced locally within the neurons of the ENS itself. However, the sources and mechanisms of production can vary for some neurotransmitters.
Following table lists a partial list of the neurotransmitters used by the ENS. The intricate interplay of these chemical messengers allows the ENS to control the complex functions of the digestive system, from breaking down food to regulating immune responses. Research is ongoing to understand the precise roles of these neurotransmitters and how they contribute to gut health and overall well-being.
|
Neurotransmitter
|
Function in the ENS
|
Location of Synthesis
|
|
Acetylcholine (ACh)
|
Stimulates muscle contractions, increases secretions
|
Produced by enteric neurons
|
|
Norepinephrine (NE)
|
Generally inhibitory, reduces gut motility and secretions
|
Sympathetic nerve fibers
|
|
Serotonin (5-HT)
|
Regulates intestinal movements, signals pain and nausea
|
Enterochromaffin cells in the gut
|
|
Dopamine (DA)
|
Modulates gastrointestinal motility and secretion
|
Enteric neurons and other gut cells
|
|
Gamma-aminobutyric acid (GABA)
|
Typically inhibitory in CNS, less understood in ENS
|
Produced by enteric neurons
|
|
Substance P
|
Mediates pain perception, regulates motility and secretion
|
Produced by enteric neurons
|
|
Vasoactive Intestinal Peptide (VIP)
|
Causes muscle relaxation, stimulates secretion
|
Produced by enteric neurons
|
|
Nitric Oxide (NO)
|
Major inhibitory neurotransmitter, relaxes smooth muscle
|
Produced by enteric neurons
|
|
Peptide YY (PYY)
|
Reduces motility post-feeding, enhances nutrient absorption
|
Produced by endocrine cells in the gut
|
|
Glutamate
|
Excitatory neurotransmitter, involved in signaling
|
Produced by enteric neurons
|
|
Somatostatin
|
Inhibits gastrointestinal hormone secretion
|
Produced by enteric neurons
|
The enteric nervous system (ENS) does not have a barrier exactly like the blood-brain barrier (BBB). The BBB is a highly selective barrier that tightly regulates the passage of substances between the bloodstream and the brain, protecting the brain from harmful substances.
The ENS, on the other hand, is located within the wall of the gastrointestinal (GI) tract and is in close contact with the contents of the gut lumen. This means that the ENS is exposed to a wide variety of substances, including nutrients, microbes, and toxins.
However, there are several mechanisms that help to protect the ENS from harmful substances:
- Intestinal Epithelial Barrier: The intestinal epithelium forms a physical barrier between the gut lumen and the underlying tissues, including the ENS. This barrier is composed of tightly connected cells that prevent the passage of most molecules and microbes.
- Mucus Layer: The intestinal epithelium is covered by a layer of mucus that acts as an additional barrier against harmful substances. Mucus also contains antimicrobial peptides and immunoglobulins that help to fight infection.
- Immune Cells: The gut is home to a large population of immune cells that monitor the intestinal environment and respond to threats. These immune cells can recognize and eliminate harmful microbes and toxins, helping to protect the ENS.
- Enteric Glia: These cells are similar to the astrocytes in the brain that contribute to the BBB. Enteric glia play a role in maintaining the integrity of the intestinal barrier and modulating immune responses in the gut.
- Blood-Enteric Barrier: While not as selective as the BBB, there is some evidence to suggest that the blood vessels supplying the ENS have tighter junctions than blood vessels in other parts of the body. This may help to limit the passage of some substances from the bloodstream into the ENS.
In summary, while there is no barrier between the ENS and blood that is directly comparable to the blood-brain barrier, there are several protective mechanisms in place to ensure the proper functioning and protection of the ENS.
Is there any well known list of chemicals impermeable(NOT crossing) the barrier(blood-ENS barrier) ?
There isn't a well-known, comprehensive list of chemicals specifically for the blood-ENS barrier like the blood-brain barrier (BBB). The blood-ENS barrier, while sharing some similarities with the BBB, is not as tightly regulated and studied. The focus of research has been primarily on the BBB due to its critical role in protecting the brain.
However, we can infer some substances that likely have limited passage across the blood-ENS barrier based on their properties and the mechanisms involved:
- Large molecules: Molecules with high molecular weight, like proteins, peptides, and complex carbohydrates, generally have difficulty crossing the blood-ENS barrier due to the tight junctions between endothelial cells.
- Hydrophilic molecules: Water-soluble molecules, like charged ions and polar compounds, typically have limited permeability across the lipid-rich cell membranes that make up the barrier.
- Highly charged molecules: Substances with strong positive or negative charges often face challenges passing through the barrier due to electrostatic interactions with the cell membrane.
- Certain drugs: Some medications, particularly those designed to target the central nervous system, are specifically engineered to not cross the BBB and may have similar limitations at the blood-ENS barrier.
- Pathogens and Toxins: Most pathogens and toxins are too large or hydrophilic to readily cross the blood-ENS barrier. However, some may have mechanisms to breach this barrier, either by damaging the intestinal epithelium or using specific transporters.
It's important to note that the blood-ENS barrier is not completely impermeable, and certain substances can still cross it through various mechanisms, such as:
- Passive diffusion: Small, lipid-soluble molecules can pass through the cell membranes of the barrier.
- Active transport: Specific transporters can carry certain molecules across the barrier.
- Receptor-mediated transcytosis: Some molecules can bind to receptors on the endothelial cells and be transported across the barrier.
The permeability of the blood-ENS barrier can also be affected by factors like inflammation and disease states. More research is needed to fully understand the transport mechanisms and selectivity of this barrier.
The development of the enteric nervous system (ENS) is a complex and highly orchestrated process that begins early in embryonic development. The ENS originates from cells called neural crest cells, which are a transient group of cells unique to vertebrates that arise from the dorsal part of the neural tube. The ENS is one of the most intricate parts of the autonomic nervous system, and its development is critical for the physiological functioning of the gastrointestinal system. The intricate
process of ENS development highlights the importance of genetic, environmental, and molecular factors in the healthy functioning of the digestive system.
1. Origin from Neural Crest Cells
- Migration: Neural crest cells destined to become the ENS begin their journey by migrating from the neural tube along specific pathways. These cells migrate into the developing gastrointestinal tract. The cells that form the ENS specifically come from the vagal and sacral regions of the neural crest.
- Proliferation: After reaching the gut, these neural crest cells proliferate extensively. They move in a rostrocaudal (head to tail) direction along the gut.
2. Colonization of the Gut
- The migrating neural crest cells colonize the entire length of the gut. The vagal neural crest cells colonize the foregut and midgut, while sacral neural crest cells contribute to the hindgut.
- This migration is crucial and must occur for the gut to be properly innervated. If cells fail to colonize any part of the gut, it results in developmental defects such as Hirschsprung’s disease, where the affected segment of the colon lacks innervation, causing severe constipation or intestinal obstruction.
3. Differentiation into Neurons and Glial Cells
- Once they have populated the gut, the neural crest-derived cells differentiate into various types of neurons and glial cells. The differentiation is influenced by interactions with the local gut environment, including factors released by the gut epithelium and mesenchyme.
- Various types of neurons are formed, each with specific neurotransmitters and functions, contributing to the diverse and complex nature of the ENS.
4. Formation of Ganglia and Plexuses
The cells organize into ganglia, which are clusters of nerve cells. These ganglia are interconnected by bundles of nerve fibers to form two main networks or plexuses within the walls of the gut:
- Myenteric Plexus (Auerbach’s Plexus): Located between the muscle layers of the gut, it primarily controls gut motility.
- Submucosal Plexus (Meissner’s Plexus): Found in the submucosa, it primarily regulates enzyme secretion and blood flow.
5. Maturation and Functional Integration
Over time, these neurons and networks mature and begin to functionally integrate with the gut’s activities. This involves forming synaptic connections and establishing communication pathways both within the ENS and with the central nervous system.
6. Regulatory Factors
The development of the ENS is regulated by a variety of genetic and molecular signals. Important genes include those encoding for the RET proto-oncogene, the endothelin receptor type B, and various others that guide migration, differentiation, and survival of neural crest cells.
The enteric nervous system (ENS) and the central nervous system (CNS) have a bidirectional communication system often referred to as the gut-brain axis. This complex interaction allows for the coordination of various gastrointestinal functions and influences overall health and well-being.
What are Key pathways of interaction ?
- Neural Pathways:
- Vagus Nerve: The vagus nerve is the primary pathway for communication between the ENS and the CNS. It carries sensory information from the gut to the brain and transmits signals from the brain to regulate gut functions like motility, secretion, and blood flow.
- Spinal Cord: The ENS also communicates with the CNS through the spinal cord, primarily via sympathetic and parasympathetic nerves. These nerves control various aspects of gut function, including pain perception and immune responses.
- Hormonal Signaling:
- The gut produces various hormones, such as serotonin, ghrelin, and cholecystokinin, that can influence the CNS by acting on specific receptors. These hormones play roles in regulating mood, appetite, and satiety.
- Immune System:
- The ENS interacts with the gut's immune system, which can influence the CNS through the release of cytokines and other inflammatory molecules. This communication pathway is implicated in various conditions, including irritable bowel syndrome (IBS) and anxiety.
- Microbiota:
- The gut microbiota, the vast community of microorganisms residing in the gut, can influence the CNS through various mechanisms, including the production of neurotransmitters and metabolites. This interaction is believed to play a role in mood regulation, stress response, and even neurological disorders.
What are Key functions of ENS-CNS interaction ?
- Gut Motility and Secretion: The CNS coordinates gut motility and secretion patterns based on the information received from the ENS. This ensures efficient digestion and nutrient absorption.
- Satiety and Appetite: The gut hormones signal the CNS about fullness and hunger, regulating food intake and energy balance.
- Pain Perception: The ENS transmits pain signals to the CNS, which then initiates appropriate responses to protect the gut from damage.
- Stress Response: The gut-brain axis plays a significant role in the body's response to stress. Stress can affect gut function, and conversely, gut problems can exacerbate stress.
- Mood and Mental Health: The gut microbiota and its interaction with the ENS are increasingly recognized as important factors in mood regulation and mental health conditions like anxiety and depression.
Understanding the complex interactions between the ENS and CNS is crucial for developing new therapies for various gastrointestinal and neurological disorders. Research in this area is ongoing and rapidly evolving, with the potential to revolutionize our understanding of the gut-brain connection and its impact on human health.
Are there any direct synaptic connection between ENS and CNS ?
No, there are no direct synaptic connections between the neurons of the enteric nervous system (ENS) and the central nervous system (CNS). The ENS is sometimes referred to as the "second brain" due to its complexity and ability to function independently to a certain degree. However, it communicates with the CNS through indirect pathways, primarily via the vagus nerve.
The vagus nerve is a mixed nerve containing both sensory and motor fibers. Sensory fibers from the ENS transmit information about the state of the gut to the CNS, while motor fibers from the CNS transmit signals to modulate gut function. These signals are relayed through interneurons in the brainstem and other regions of the CNS.
In addition to the vagus nerve, the ENS also communicates with the CNS through the spinal cord via sympathetic and parasympathetic pathways. These pathways play a role in regulating gut motility, secretion, blood flow, and immune responses.
While there are no direct synaptic connections, the communication between the ENS and CNS is extensive and complex, allowing for coordinated regulation of gut function and overall bodily homeostasis. This bidirectional communication is often referred to as the gut-brain axis.
How the chemicals and metabolites can pass the barrier to mediate the interaction between ENS and CNS ?
While the blood-ENS barrier does limit the passage of certain substances, it is not completely impermeable, and various mechanisms allow for the transport of chemicals and metabolites necessary for communication between the ENS and CNS:
- Diffusion:
- Small, lipid-soluble molecules: Some signaling molecules, such as certain neurotransmitters (e.g., nitric oxide) and microbial metabolites (e.g., short-chain fatty acids), are small and lipophilic enough to passively diffuse across the endothelial cell membranes of the blood-ENS barrier.
- Active Transport:
- Specific transporters: The blood-ENS barrier expresses various transporters that actively shuttle specific molecules across the barrier. For example, glucose transporters facilitate the uptake of glucose into the ENS, which is essential for its energy metabolism.
- Efflux pumps: These transporters actively pump certain substances out of the ENS and back into the bloodstream, preventing their accumulation within the enteric nervous tissue.
- Receptor-mediated Transcytosis:
- Larger molecules: Some larger molecules, such as cytokines and certain neuropeptides, can bind to specific receptors on the endothelial cells. This binding triggers a process called receptor-mediated transcytosis, where the molecule is engulfed by the cell and transported across the barrier in vesicles.
- Paracellular Transport:
- Tight junctions: While tight junctions between endothelial cells limit the passage of most substances, they are not completely sealed. Small gaps between the cells can allow for the paracellular transport of certain molecules, especially during inflammation or increased gut permeability.
- Immune Cell Trafficking:
- Immune cells: Immune cells can migrate across the blood-ENS barrier, carrying signaling molecules and interacting with both the ENS and the CNS. This is crucial for maintaining gut homeostasis and regulating immune responses.
Overall, the blood-ENS barrier is a dynamic and selective barrier that allows for the controlled passage of specific molecules necessary for the communication between the ENS and CNS. While it limits the entry of potentially harmful substances, it also facilitates the transport of essential nutrients, signaling molecules, and immune cells, ensuring the proper functioning and coordination of the gut-brain axis.
Reference
YouTube/Other Video
|
|

